Ionic Bond
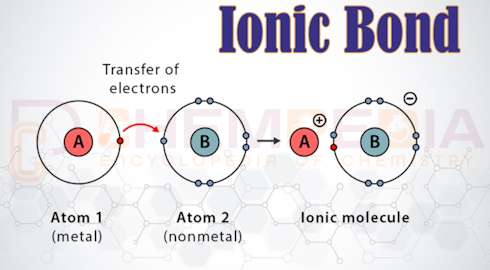
Ionic Bond Definition An ionic bond (electrovalent bond) is the electrostatic force of attraction between oppositely charged ions formed by the complete transfer of one or more electrons from one atom to another. This concept was first explained by Walther Kossel and Gilbert N. Lewis in 1916. Usually: Metals lose electrons → cations Non-metals gain [...]